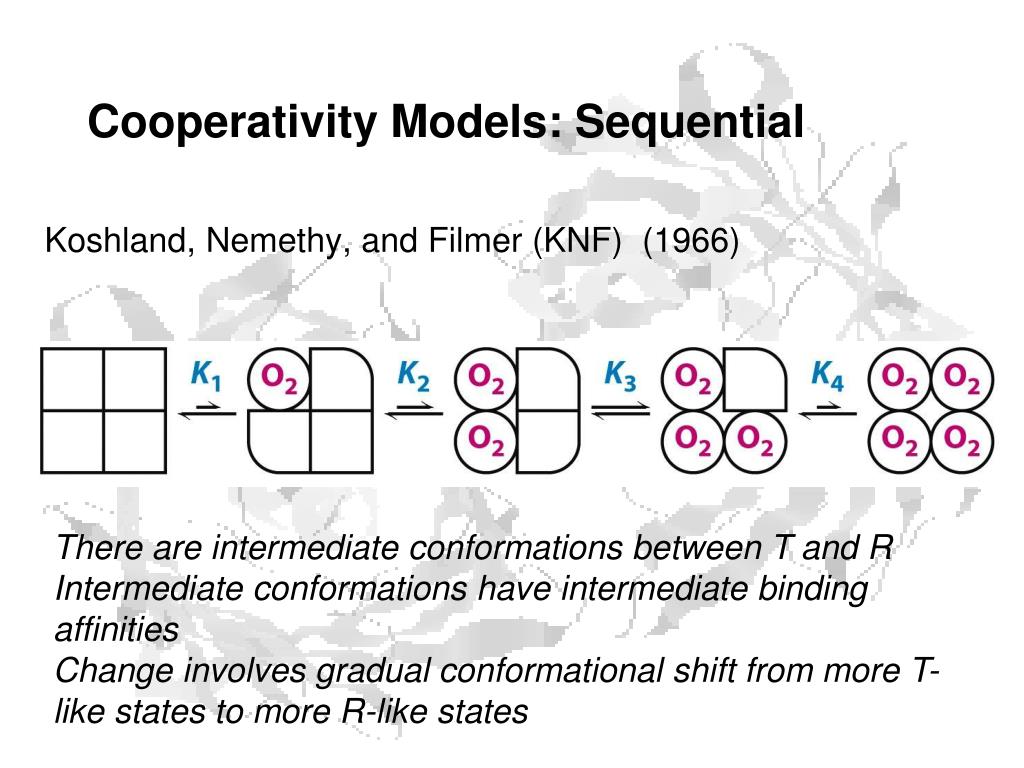
Treatment of other geometries leads to the same expressions for the concentrations except for the subunit interaction terms, which are different for each geometry. _ KSKt KAB +3KsKt2KABKBB2+3KSKt3KABKBB3 + KsKfKBB4 Treatment of the tetrahedral tetrameric protein gives rise to the following expression: The binding to conformation A does not exist.īecause the sequential model deals with subunit interactions explicitly and refers to contacts between subunits, it is necessary to consider the geometry of the molecule for example, a tetrameric protein may exist as square or tetrahedral of which tetrahedral is the preferred arrangement. Ligand binds only to conformation B, with association constant Ks. The stability of any particular state is determined by a product of equilibrium constants including a term Kt for each subunit in the B conformation expressing the free energy of the conforma-tional transition from A to B of an isolated subunit, a term K for each contact between a subunit in the A conformation with one in the B conformation, and a term KBB for each contact between a pair of subunits in the B conformation.ģ. There is no requirement for conformational symmetry therefore mixed conformations are permitted. Each subunit in the protein is capable of existing in either of two conforma-tional states A and B.Ģ. The model is based on three postulates:ġ. The sequential model proposes that the conformational stability of each subunit is determined by the conformations of the subunits with which it is in contact. The sequential model of Koshland, Nemethy and Filmer (Koshland et al., 1966). = a(1 + ay-1 + LCa(1 + a)n - V = (1 + a)n + L (1 + a)n where a = SKr and Ca = SKT. The fractional saturation has the following expression: The dissociation constants, KT and Kr for binding of ligand S to protomers in the T and R conformations, respectively, are different, with the ratio Kr/Kt = C that is assumed to favor binding to the R conformation. Thus only a single equilibrium constant L = / is sufficient to express the equilibrium between them.ģ. Therefore, a protein with n subunits is limited to only two conformational states, Tn and Rn. The conformational symmetry is maintained such that all protomers of the protein must be in the same conformation, either all T or all R at any instance. Each subunit (protomer) in the protein is capable of existing in either of two conformational states, namely, T (tight) and R (relaxed) states.Ģ. The model is based on three postulates about the structure of an oligomeric protein (allosteric protein) capable of binding ligands (allosteric effectors):ġ. This model was originally termed the allosteric model. The symmetry model of Monond, Wyman, and Changeux (Monad et al., 1965). Two models have been proposed for the cooperativity of homotropic interactions between ligand and proteins: Furthermore, the cooperativity can be observed with interactions of a single kind of ligand in homotropic (same ligand) interactions or those involving two (or more) different kinds of ligands in heterotropic interactions. Although there is no need for such proteins to be oligomeric, nearly all known cases of cooperativity at equilibrium are found in proteins with separate binding sites on different subunits. The cooperativity refers to interaction between binding sites in which the binding of one ligand modifies the ability of a subsequent ligand molecule to bind to its binding site, whereas the allosterism refers to the binding of ligand molecules to different sites.īecause a single binding site cannot generate cooperativity, a cooperative binding system must consist of two or more binding sites on each molecule of protein. The interaction that causes an increase in successive association constants is called positive cooperativity, while a decrease in successive association constants is the consequence of negative cooperativity. The observed changes in successive association constants for a multiple ligand binding system suggest the cooperativity in the interaction between binding processes. The concepts of cooperativity and allosterism (Koshland et al., 1966 Monad et al., 1965 Richard and Cornish-Bowden, 1987) in proteins have been applied to explain various ligand-receptor interaction phenomena. The ligand-induced conformational changes appear to be an important feature of receptors with regulatory functions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
